News
Outside panel advises FDA to approve Pfizer RSV vaccine for older adults
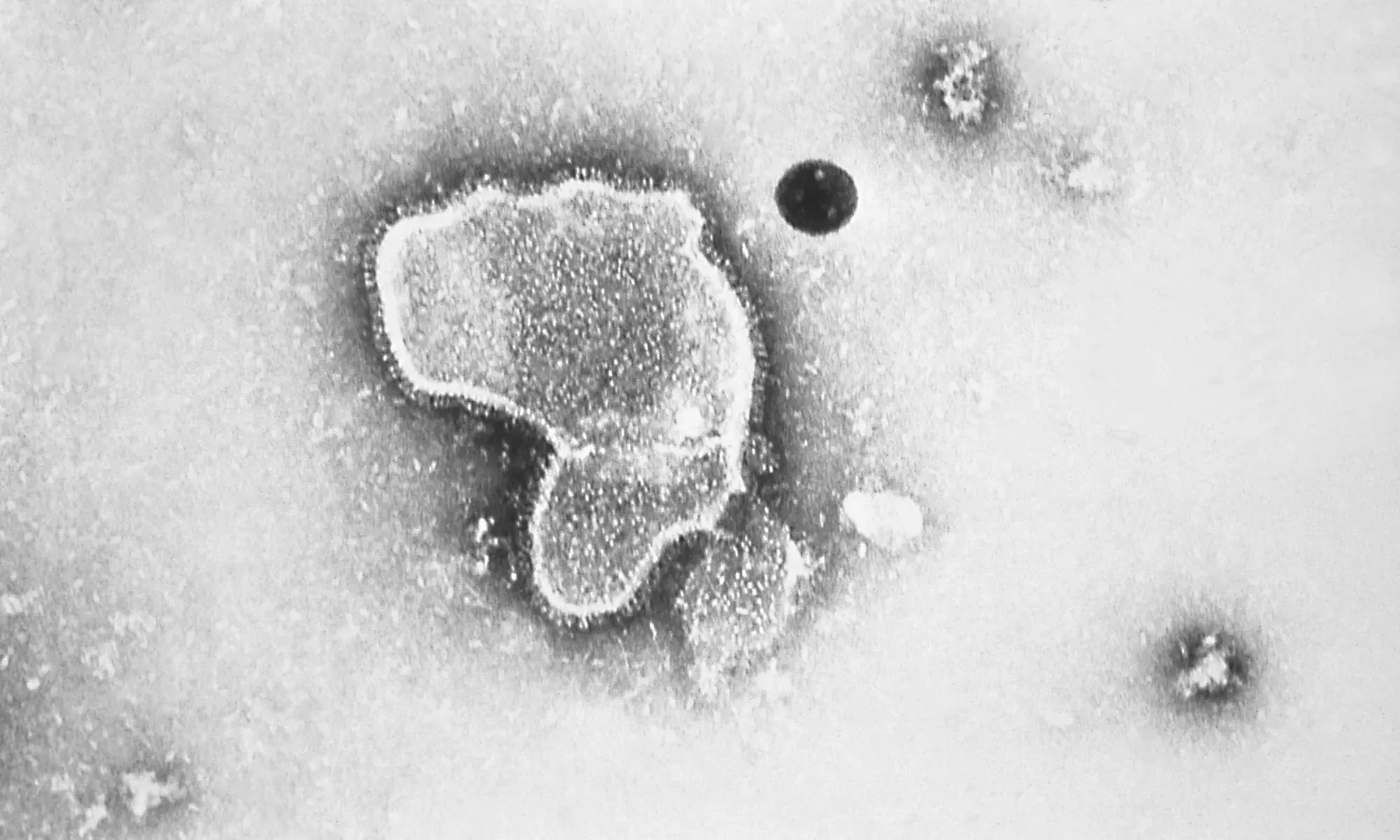
An external panel that advises the United States Food and Drug Administration narrowly voted to endorse a new Pfizer vaccine for respiratory syncytial virus (RSV) for older adults.
The move is the first step toward the FDA approving a vaccine that prevents severe RSV in the lower respiratory systems of adults aged 65 and older. While RSV typically leads to cold or flu-like symptoms that resolve within a few days, for older people and infants it can be severe, sometimes requiring hospitalization. The virus results in 60,000-160,000 hospitalizations among adults 65 years and older per year in the United States, according to the Centers for Disease Control and Prevention.
The 7-4 vote confirmed the panel’s opinion that sufficient data exists to prove the vaccine is safe and effective at preventing RSV in older adults. One panel member abstained from the Wednesday vote. The votes against approving the vaccine were heavily based on some instances of Guillain-Barre syndrome appearing within days of taking the vaccine in trials. Two adults in Pfizer’s trials experienced GBS after taking the RSV vaccine.
Guillain-Barre syndrome, according to the CDC, is a bodily response that occurs when the immune system begins harming the nerves. It can lead to muscle weakness, and in some cases, paralysis. The same advisory panel also voted this week to advance an RSV vaccine manufactured by GlaxoSmithKline (GSK) meant for individuals 60 years and older.
Terry A. Hurlbut has been a student of politics, philosophy, and science for more than 35 years. He is a graduate of Yale College and has served as a physician-level laboratory administrator in a 250-bed community hospital. He also is a serious student of the Bible, is conversant in its two primary original languages, and has followed the creation-science movement closely since 1993.
-

 Guest Columns4 days ago
Guest Columns4 days agoDressed To Kill: Women of the Left and the Cult of Violence Worship
-

 Executive3 days ago
Executive3 days agoWaste of the Day: Iraq Training Money Squandered
-

 Civilization2 days ago
Civilization2 days agoGOP Battles Sharia: Is Islamic Law a Threat or Dog Whistle?
-

 Civilization3 days ago
Civilization3 days agoPope Just Gave US, Allies the Strategic Edge Over Beijing
-

 Accountability4 days ago
Accountability4 days agoUS Declaration at 250: New Challenges, Enduring Principles
-

 Executive3 days ago
Executive3 days agoWaste of the Day: Viagra for the Military
-

 Civilization1 day ago
Civilization1 day agoCornyn’s Defeat and the Remaking of the GOP
-

 Civilization3 days ago
Civilization3 days agoSlavery, the Underground Railroad, and the Civil War


